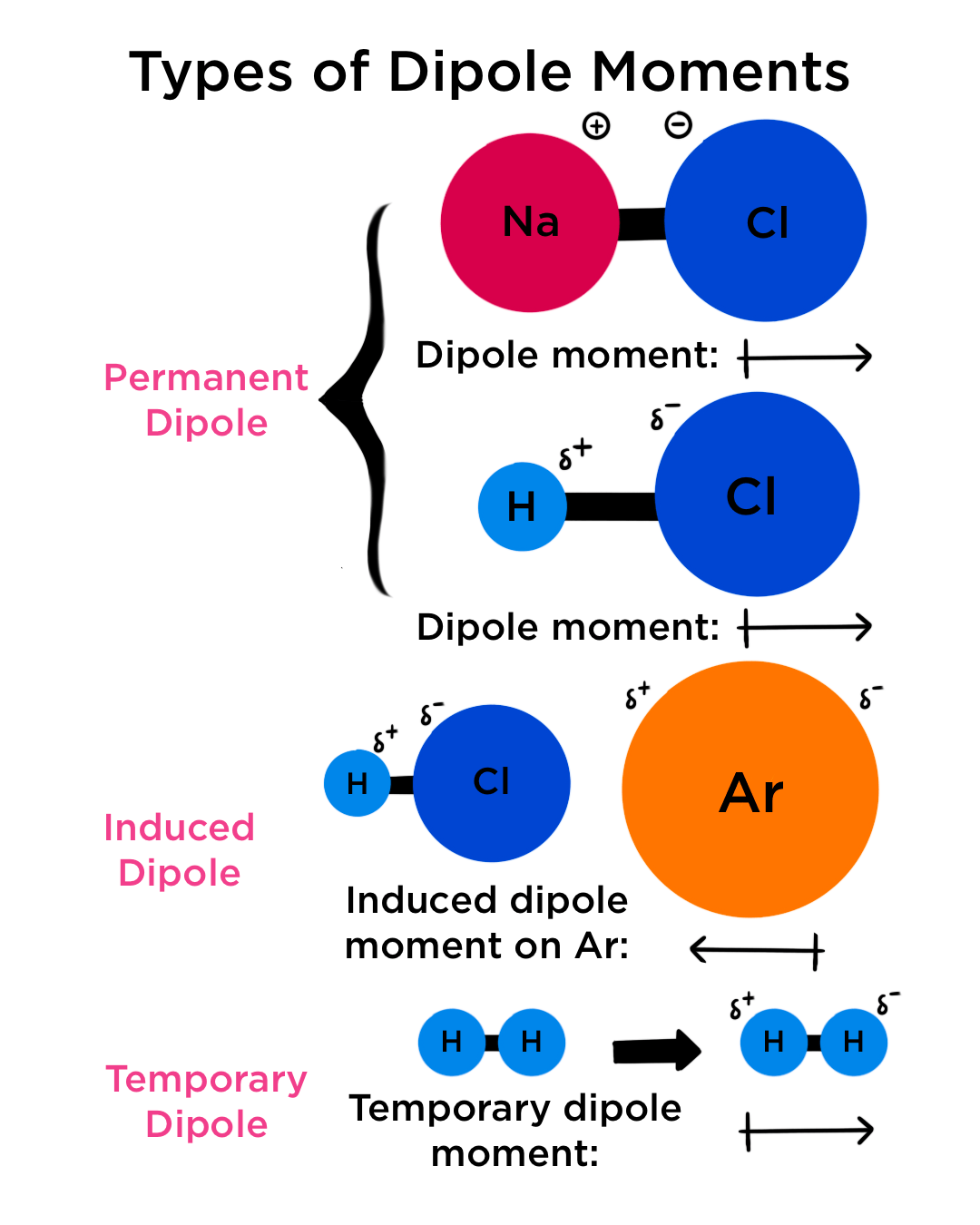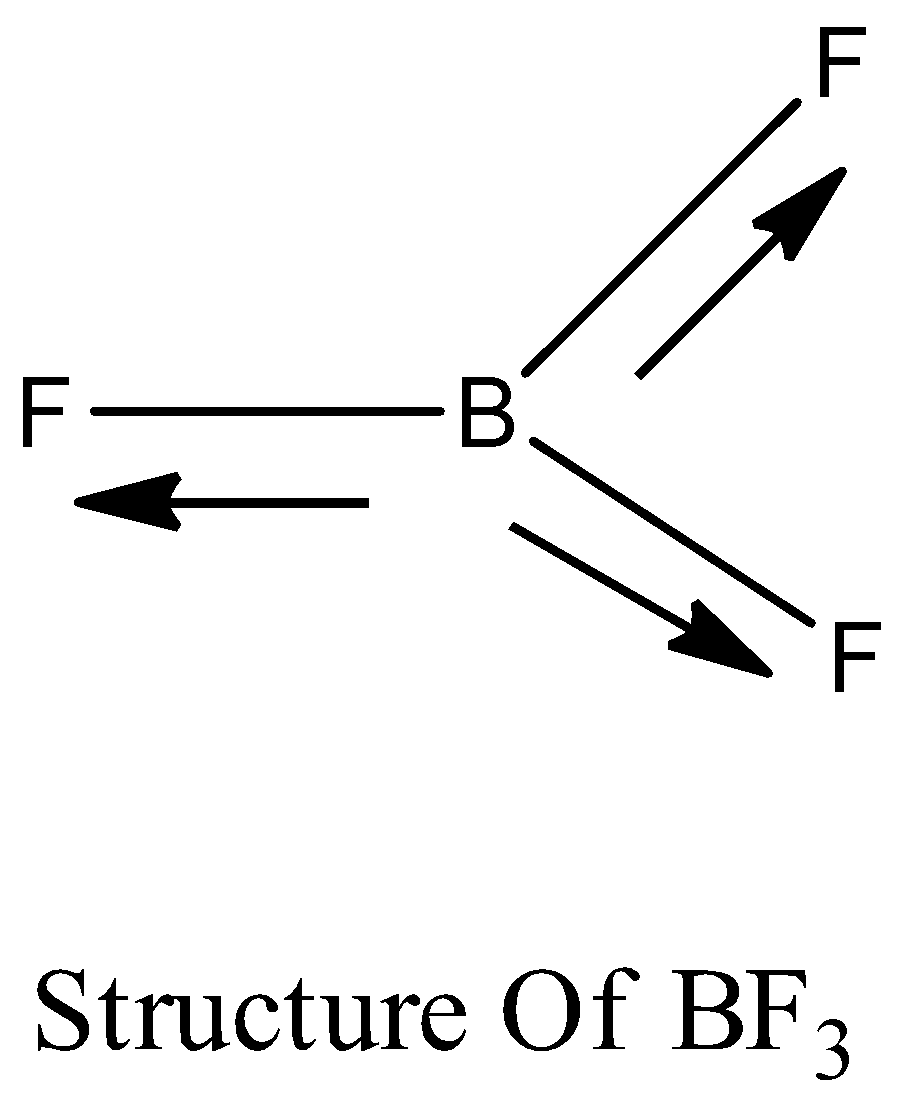
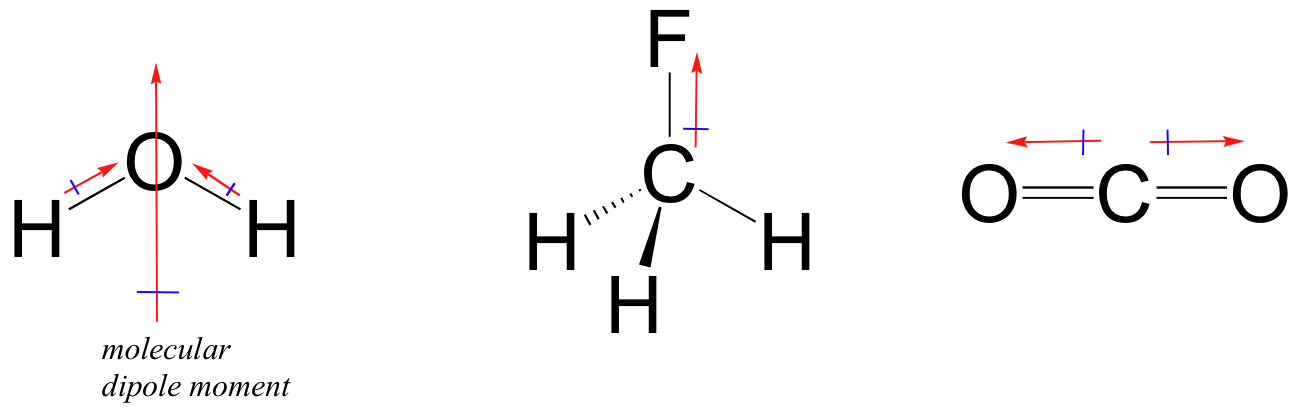
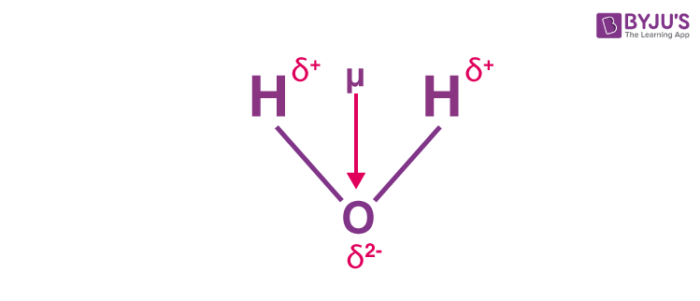
Boron trifluoride $(B{F_3})$ has no dipole moment $(\\mu = 0D)$. Explain how this observation confirms the geometry of $B{F_3}$ predicted by VSEPR theory.
Dipole moment curves of NO (left) and AlO (right). The vertical dashed... | Download Scientific Diagram

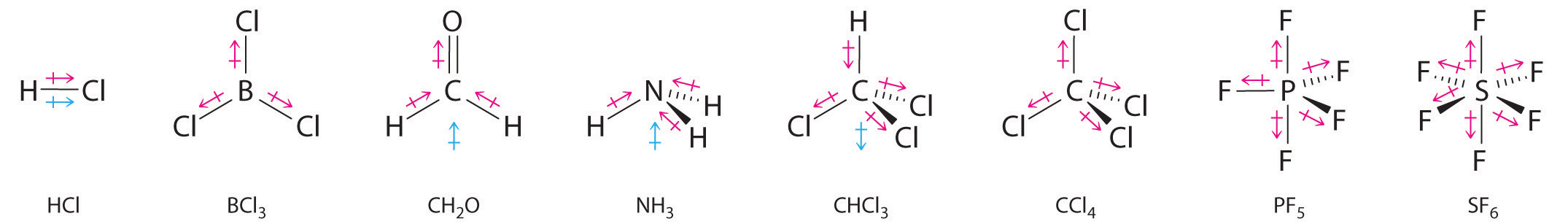
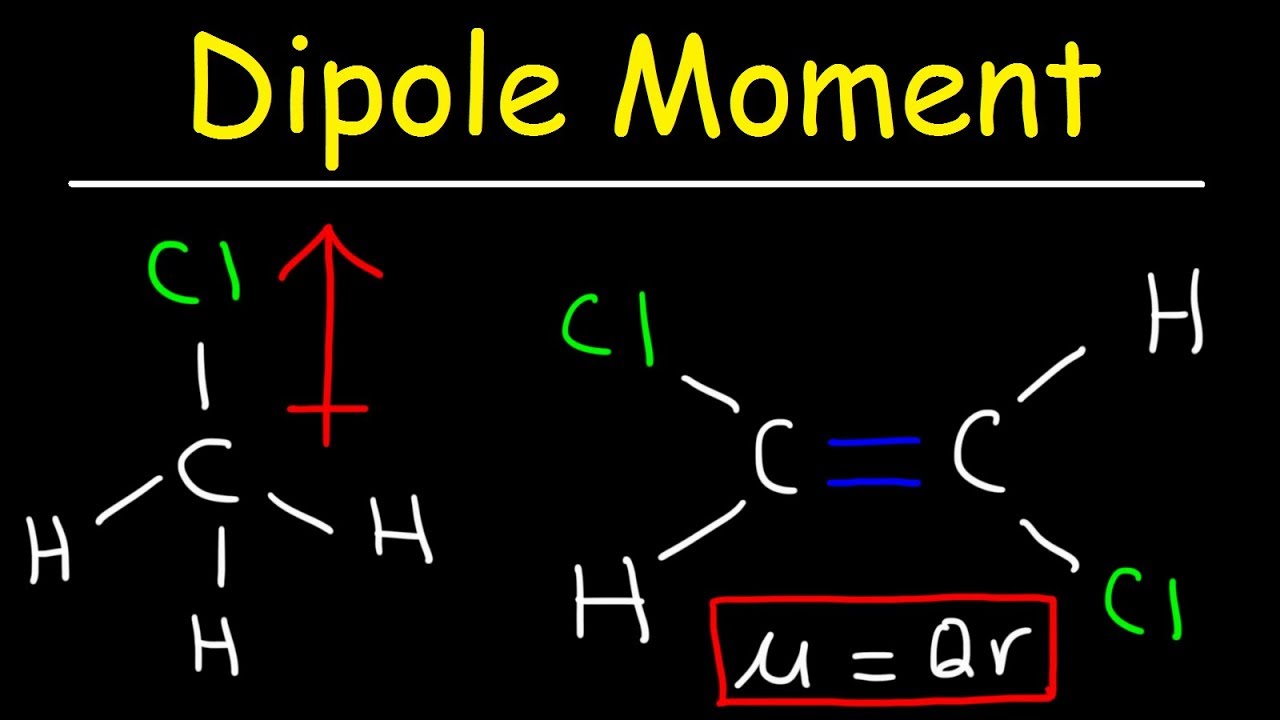




/chapter3/pages3and4/page3and4_files/cnodipole.png)

/chapter3/pages3and4/page3and4_files/cnoorbital.png)