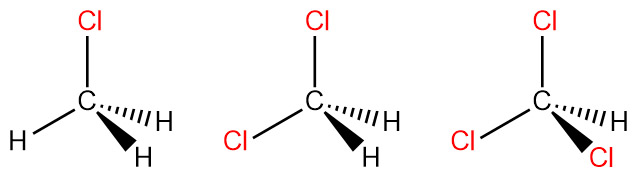
Welcome to Chem Zipper.com......: The Dipole moment of chloromethane is more than fluoromethane. Explain.

Is the molecule CH3Cl polar or nonpolar? If it is polar, specify the direction of its polarity. | Homework.Study.com

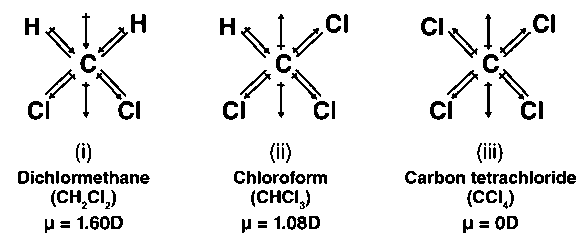
Please explain the dipole moment of CH2Cl2,CH3Cl,CCl4,CHCl3 using diagram( direction )and arrange in increasing order of dipole moment - Chemistry - Haloalkanes and Haloarenes - 16270361 | Meritnation.com

halides - Why does chloromethane have a larger dipole moment than chloroform? - Chemistry Stack Exchange
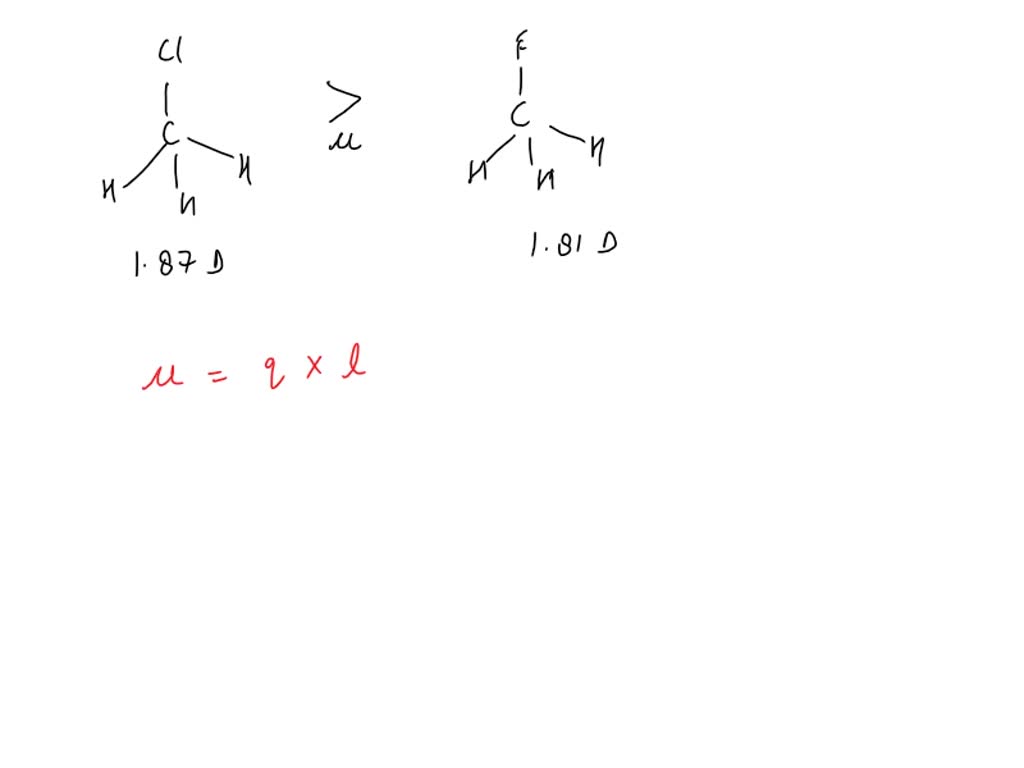
SOLVED: Fluoromethane (CH3F, μ=1.81 D) has a smaller dipole moment than chloromethane (CH3Cl, μ=1.87 D) even though fluorine is more electronegative than chlorine. Explain.
When we compare the dipole moment of CH3Cl, CH2Cl2 and CHCl3 we see that CH3Cl has the greatest value (which is greater than water too).Why does this happen? - Quora

Why the dipole moment of CH3F is less than CH3Cl although 'F' is more electronegative than 'Cl' ? - CHEMSOLVE.NET | In this moment, Bond length, Chemistry
![Best Answer] Which has highest dipole moment?? CH3Cl, CH2Cl2, CHCl3 or CCl4. well, I saw a video or Merit - Brainly.in Best Answer] Which has highest dipole moment?? CH3Cl, CH2Cl2, CHCl3 or CCl4. well, I saw a video or Merit - Brainly.in](https://hi-static.z-dn.net/files/df7/fd9f40d6eb64e74ec8cead9b4608b313.jpg)
Best Answer] Which has highest dipole moment?? CH3Cl, CH2Cl2, CHCl3 or CCl4. well, I saw a video or Merit - Brainly.in
✓ Solved: What is a dipole moment ? Give four examples of molecules that possess dipole moments, and...
Why does CH3Cl have more dipole moment than CH2Cl2 though by vector addition CH2CL2 should have more? - Quora
Compare the dipole moment between CHCl_3 and CH_3Cl. Why dipole moment of CH_3Cl is more than that of CHCl_3.
Which of the following molecules has no dipole moment? (a) CH3Cl (b) CHCl3 (c) CH2Cl2 (d) CCl4 - Sarthaks eConnect | Largest Online Education Community