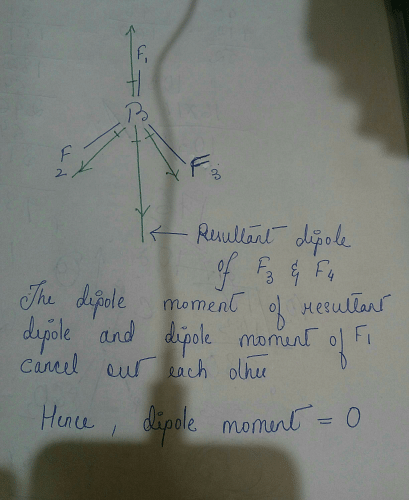
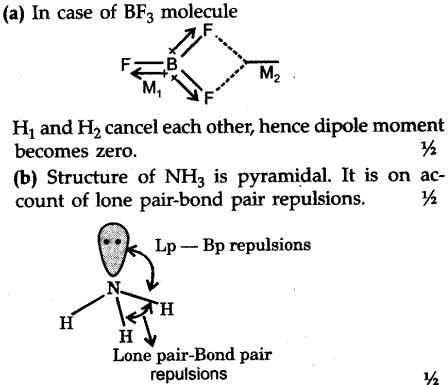
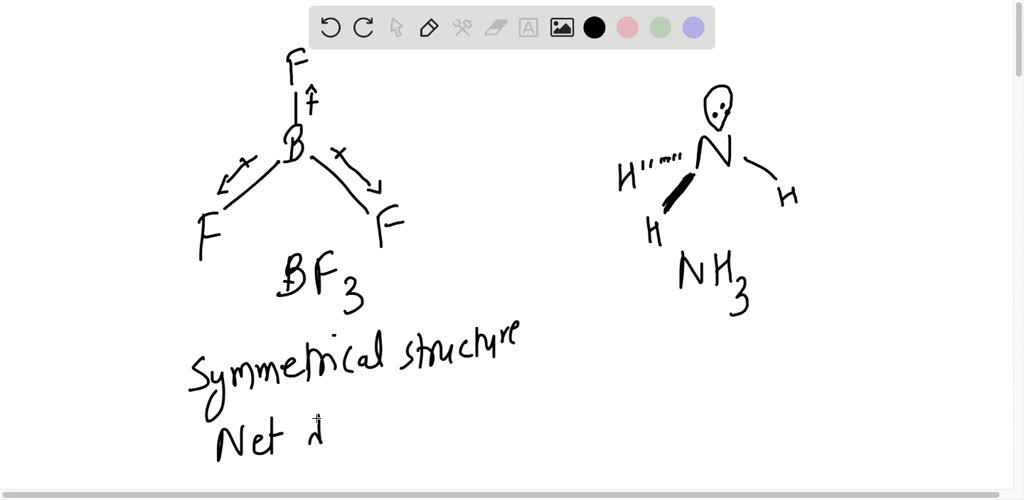
Question 17 Give reason the following: @ Dipole moment of BF3 is zero but ammonia has a dipole moment. (6) Cuci is covalent than Naci. C) LiCl is covalent than NaCl. (

why does BF3 has a zero dipole moment although the B-F bondsare polar - Chemistry - Chemical Bonding and Molecular Structure - 10846009 | Meritnation.com
Which out of the following pairs has dipole moment and why ? nbsp; i] BF3 AND NF3 nbsp; ii] CO2 AND H2O
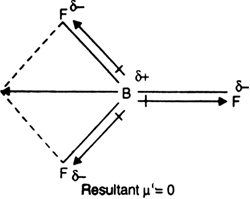
Dipole moment in case of BF3 is zero. Explain. - Sarthaks eConnect | Largest Online Education Community
Which out of the following pairs has dipole moment and why ? nbsp; i] BF3 AND NF3 nbsp; ii] CO2 AND H2O
Define dipole moment? Comment on structure & dipole moment of CO2, BF3. - Sarthaks eConnect | Largest Online Education Community

Compare dipole moment in BF3 and BF4- - Chemistry - Chemical Bonding and Molecular Structure - 10695965 | Meritnation.com

![Odia] Among C Cl4, BF3, NH3 and CO2, Which one has net dipole moment Odia] Among C Cl4, BF3, NH3 and CO2, Which one has net dipole moment](https://d10lpgp6xz60nq.cloudfront.net/ss/web-overlay-thumb/11812254.webp)