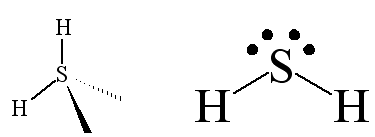
Calculations of dipole moments of H2S and PH3 - Transactions of the Faraday Society (RSC Publishing)

90 DUT8U J U TU Dipole moment of H2S is 0.95 D. Calculate the bond moment the bond angle is 97" (Cos 48.5° = 0.662) 0.7170 thi 1 275 Å The percentage of one

How to Calculate the Strength of Intermolecular Forces between Different Molecules from Chemical Structure | Chemistry | Study.com

Explain the difference in the solubility of water and hydrogen sulfide in hexane? Solubility of water in hexane is 0.01 g/100g Solubility of hydrogen sulfide in hexane is 0.7g/100g? | Socratic

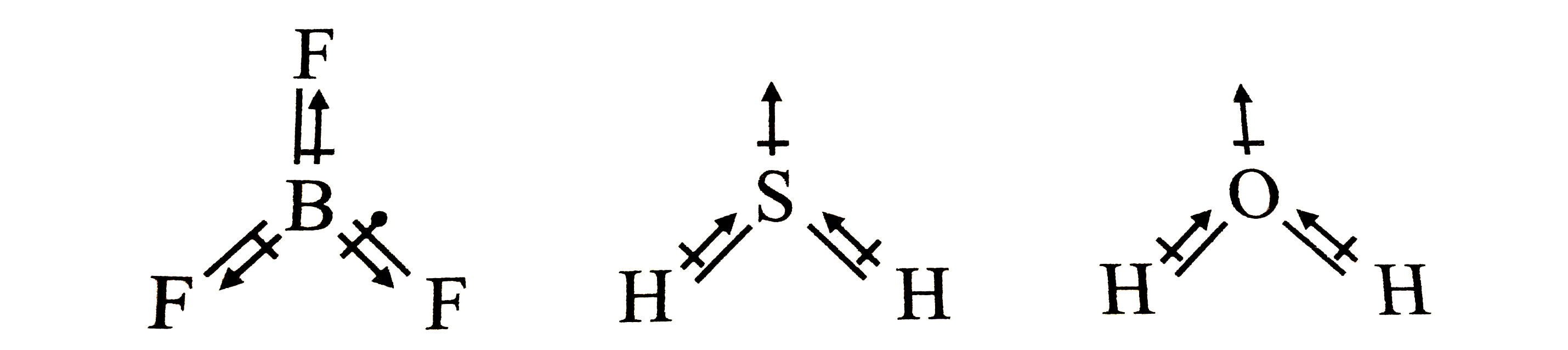
Which of the following molecules does not possess a permanent dipole moment a)H2S b)SO2 c)SO3 d)CS2 - Chemistry - The p-Block Elements - 12272413 | Meritnation.com

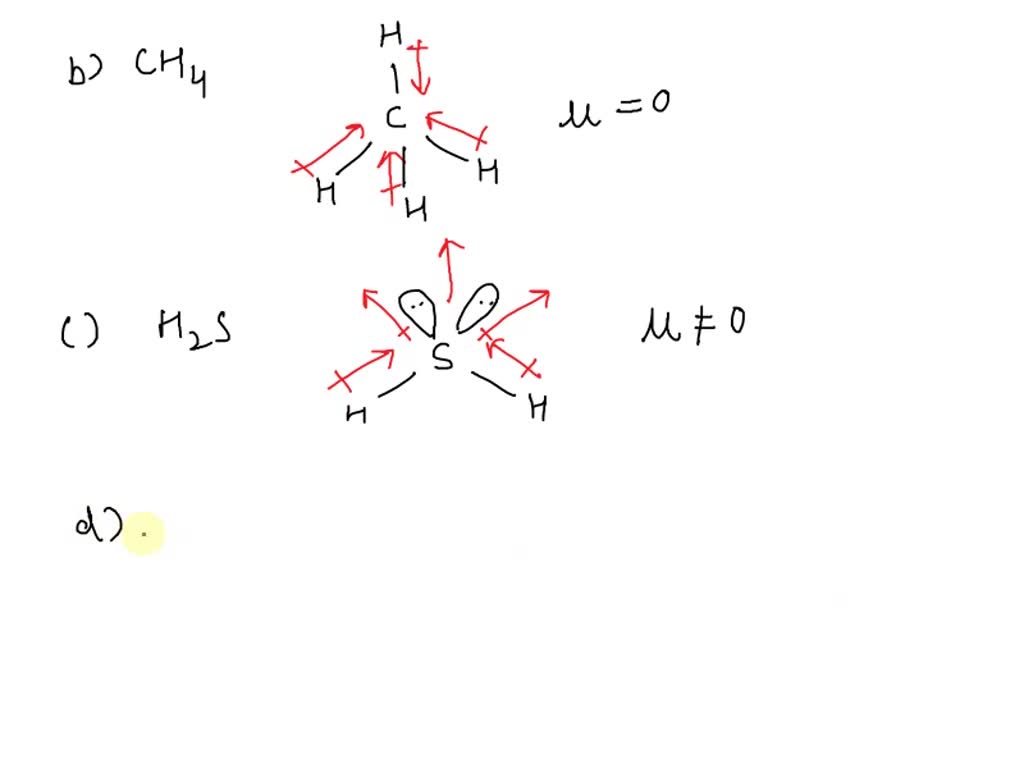
Question The geometry of H2S and its dipole moment are: A. angular and non-zero B. angular and zero C. linear and non-zero D. linear and zero

SOLVED: which of the following is polar (possesses a dipole moment) H2S C6H6 I2 CH4 None of the above






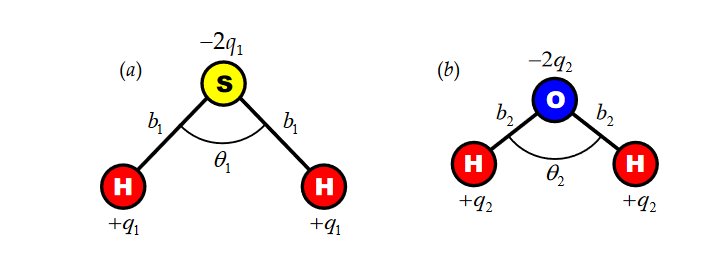

![PDF] The dipole moment surface for hydrogen sulfide H2S | Semantic Scholar PDF] The dipole moment surface for hydrogen sulfide H2S | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/0ffa91c8852350858908b29f68b54676d71c7906/7-Table3-1.png)